Identification of Targets for Cancer Treatment
Generation of small molecule libraries by diversity-oriented synthesis (DOS) may reveal the expression of proteins by genes and thereby provide new targets for pharmaceutical treatment.
Cancer and several other severe illnesses are related to genetics. Of interest here are not only the genes themselves but even more so the expression of their products, i.e. proteins. The field is called epi-genetics. A sub-field is reverse chemical genetics in which small molecules are screened against isolated proteins in order to modulate the protein function. The thesis presents novel strategies for synthesis of compound collections for this purpose.
The publication of the human genome sequence in 2001 provided scientists with the opportunity to understand the role of all genes in normal physiology as well as pathology. However, the gene sequence in itself does not provide information on the dynamic expression of proteins which is of key interest to a number of drug discovery applications.
It has been estimated that 10-14 % of the human genome encodes proteins that are likely to be modulated by existing drug-like small molecules. Nonetheless, only around 1,000 of these approximately 30,000 genes have known chemical modulators. As a consequence many human disease-related targets are currently “undruggable”.
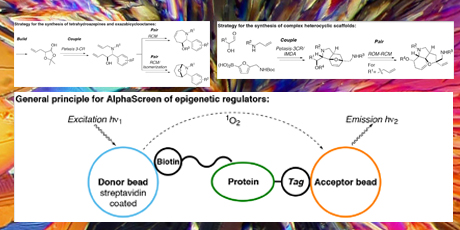
In order to identify more targets, the need for generation of structurally diverse compound collections is apparent. Diversity-oriented synthesis (DOS) aims at efficient generation of small molecule libraries with a high degree of complexity and scaffold diversity. Multi-component, tandem and cascade reactions are ideally suited for DOS since they provide complex frameworks in few steps. Compounds originating from DOS campaigns have been identified as potent and selective inhibitors of protein-protein interactions (PPIs) implicated in different types of cancers. In the project, a tandem RCM/isomerization/ nucleophilic addition reaction formed the basis for a library of medium-sized heterocycles. Further, a novel Petasis 3-CR/ IMDA reaction cascade in combination with a ROM-RCM sequence provided access to a library of complex sp3-rich heterocycles.
Efficient and high-yielding diversification reactions enabled the introduction of appendage and functional group diversity around the common scaffolds, and library members were synthesized in 5-6 operations. Target-based assays for high-throughput screening were developed for two epi-genetic regulators, namely the bromodomain-containing ATAD2 and the histone methyltransferase NSD1.
In conclusion, epi-genetic regulators are promising drug targets due to the potential of reverting malignant cell-states into more benign phenotypes. In this context, libraries of structurally diverse small molecules hold the potential of providing novel chemical probes for targets previously deemed undruggable. Ultimately such probes could lead to the treatment and prevention of severe diseases such as cancer.